APPOINT: With FABHALTA oral monotherapy, substantial Hb improvements without the need for RBC transfusions are within reach
A study of complement inhibitor–naive adults with PNH1
PRIMARY END POINT
Patients with sustained Hb increase of ≥2 g/dL* from baseline in the absence of RBC transfusions† after the 24-week core treatment period
*Assessed between Days 126 and 168.1,2
†Assessed between Days 14 and 168. Requiring RBCs refers to any patient receiving transfusions or meeting protocol-defined criteria.2,3
‡Based on observed and central laboratory data only. Adjusted mean assessed at a single Week 48 (Day 336) visit. Patients who received transfusions between Days 14 and 336 or were missing Day 336 assessment were considered nonresponders.4
§Assessed at a single Week 48 (Day 336) visit.4
‖The 'n' values reflect patients who responded based on observed data.4
Patients taking FABHALTA experienced an increase in adjusted mean Hb level of +4.29 g/dL from baseline
ADDITIONAL END POINT
The data from this additional analysis are exploratory; therefore, not subject to family-wise Type 1 error control, and presented for observation only. No formal conclusions can be made.
Patients experienced increases in mean Hb by Week 1, with increases through Week 242,3
Mean Hb levels (g/dL) through Week 24 (values within 30 days of transfusion are excluded and considered missing)
Click on image to enlarge.
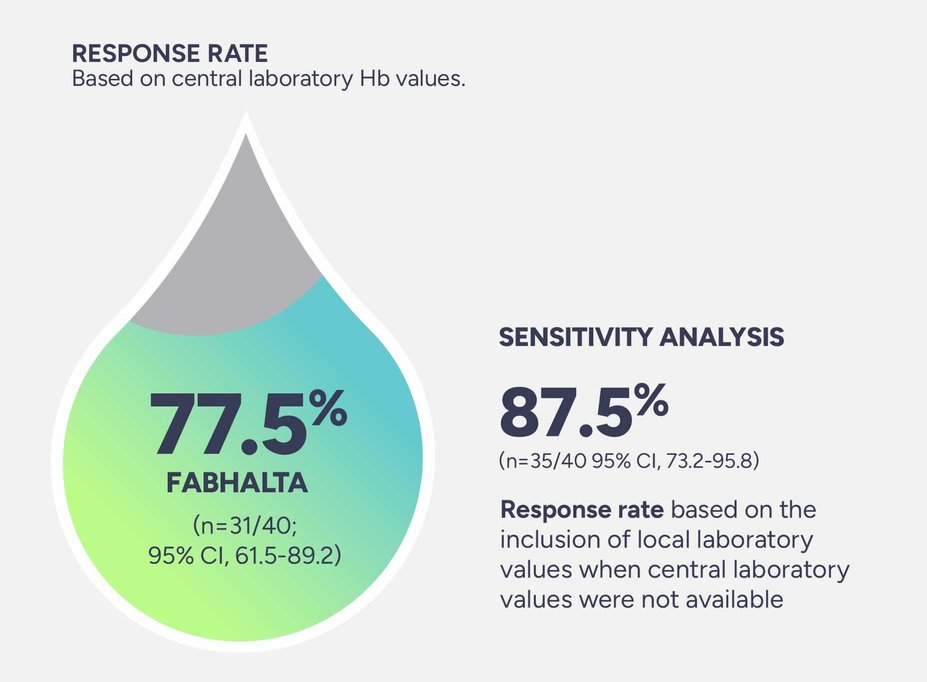
Sustained Hb levels of ≥12 g/dL* in the absence of RBC transfusions† after 24 weeks was observed in 47.5% of patients (n=19/40; 95% CI, 31.5-63.9)2
*Assessed between Days 126 and 168.1,2
†Assessed between Days 14 and 168. Requiring RBCs refers to any patient receiving transfusions or meeting protocol-defined criteria.2,3
§Assessed at a single Week 48 (Day 336) visit.4
||The ‘n’ values reflect patients who responded based on observed data.4
¶Based on central laboratory data only. Adjusted mean assessed at a single week 48 (Day 336) visit. Hb data during core treatment period and within 30 days post-transfusion were considered missing.4
#The ‘n’ values reflect patients with non-missing data.4
**Based on observed and central laboratory data only. Patients who received transfusions between Days 14 and 336 or were missing a Day 336 assessment were considered nonresponders.4
ADDITIONAL END POINT
The data from this additional analysis are exploratory; therefore, not subject to family-wise Type 1 error control, and presented for observation only. No formal conclusions can be made.
Patients achieving RBC transfusion avoidance assessed between Weeks 2 and 242
††Transfusion avoidance defined as patients who did not receive any transfusions between Days 14 and 336.4
‡‡Based on observed data considering only patients who received transfusions between Days 14 and 336 as failures.4
The impact of FABHALTA on absolute reticulocyte count (ARC) and lactate dehydrogenase (LDH)—known markers of EVH and IVH
ADDITIONAL END POINTS
The data from this additional analysis are exploratory; therefore, not subject to family-wise Type 1 error control, and presented for observation only. No formal conclusions can be made.
ARC and LDH are 2 of the known biomarkers of hemolytic activity. ARC is a marker of both IVH and EVH, while LDH primarily reflects IVH.6-8
Adjusted mean change from baseline in ARC2,3,§§
§§Mean of visits between Days 126 and 168.3
Adjusted mean change from baseline in LDH2,3,††
††Mean of visits between Days 126 and 168.3
¶¶Day 7.3
##Day 14.3
***Day 168.3
†††Adjusted mean assessed at a single Week 48 (Day 336) visit. Analysis based on observed and central laboratory data only.4
‡‡‡The ‘n’ values reflect patients with non-missing change from baseline.4
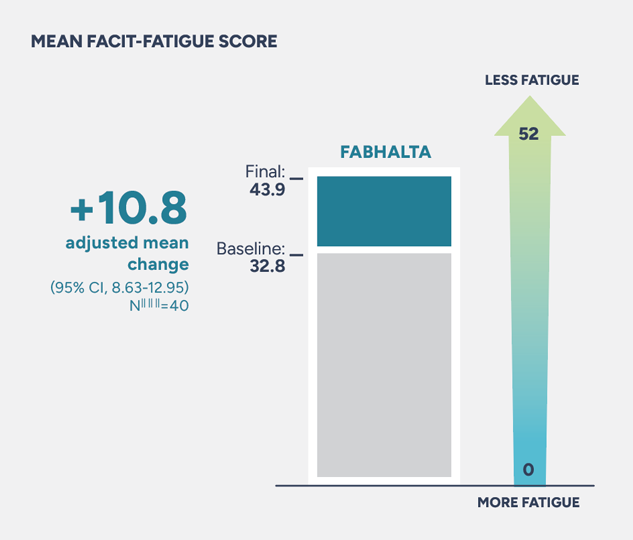
Patients who started on FABHALTA experienced an adjusted mean change of +10.8 from baseline in FACIT-Fatigue score
ADDITIONAL END POINT
Patient-reported FACIT-Fatigue scores may be an underestimation or overestimation because patients were not blinded to treatment.
The data from this additional analysis are exploratory; therefore, not subject to family-wise Type 1 error control, and presented for observation only. Due to the exploratory nature, small sample size, single-arm and open-label design, no formal conclusions can be made.
Assessed between Weeks 18 and 24. The adjusted mean change* from baseline in FACIT-Fatigue score§§§ in patients taking FABHALTA was +10.8 (95% CI, 8.63-12.95)2,3
¶¶¶The ‘n’ values reflect patients with non-missing values.4
###Adjusted mean assessed at a single Week 48 (Day 336) visit.4
In separate, large-scale surveys, the mean FACIT-Fatigue score for the general population was 449,10,****
*Assessed between Days 126 and 168.3
§§§The Functional Assessment of Chronic Illness Therapy–Fatigue Scale (FACIT-Fatigue) is a 13-item questionnaire that assesses self-reported fatigue and its impact upon daily activities and function. The level of fatigue is measured on a 4-point Likert scale (in the study, 4=not at all fatigued to 0=very much fatigued), with 0 being the worst possible score and 52 the best.3
||||||Baseline mean FACIT-Fatigue scores and adjusted mean change in FACIT-Fatigue scores at Day 168 were reported for 40 patients.3
****The FACIT-Fatigue score for the general population was determined through the assessment of 1010 adults in the US in 2002 and 2426 adults in Germany in 2018.9,10